PRESS CENTER
新闻中心
|
|
Ensheng Medical V-Mixtent ® The venous stent system made its debut at APAC and received high praise from international expertsAs an authoritative academic platform in the field of vascular surgery in the Asia Pacific region, APAC has gathered numerous top experts from home and abroad for in-depth exchanges and discussions on the latest research and clinical progress in the diagnosis and treatment of vascular diseases.
Ensheng Medical made its debut at this conference and specially invited Dr. Raden Suhartono, a renowned Indonesian vascular surgeon (hereinafter referred to as Dr. Tono), to showcase V-Mixtent together ® The innovative breakthrough of the venous stent system presents the excellent performance of this Chinese made venous stent in the treatment of venous obstructive diseases.
Ensheng Medical invited Dr. Tono to give a technical presentation at the conference. Dr. Tono's speech focused on the current situation of venous disease management in Indonesia, detailing the country's progress and challenges in the treatment of venous diseases. Indonesia faces several major challenges in the treatment of venous diseases: delayed diagnosis, limited access to advanced intravenous treatment technologies, and differences in treatment methods between urban and rural medical centers. Nevertheless, Indonesia has made significant progress with the introduction of minimally invasive technology. He highlighted the EnSheng Medical V-Mixtent in his speech ® Innovative breakthrough in venous stent and statement: "V-Mixtent ® The innovative design of venous stents has effectively solved the key technical challenges in venous therapy, and its performance is impressive.
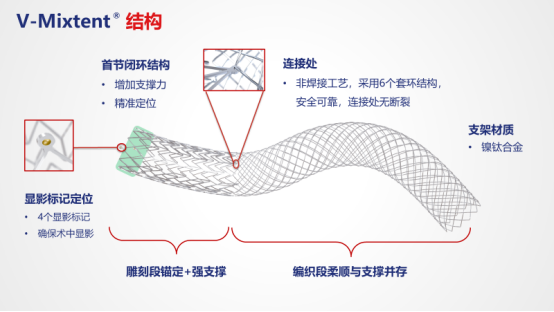
01Technological breakthrough: redefining the standard for venous stents The product adopts innovative modular design, combining strong support in carved form with flexibility in woven form. The non welded ring connection process eliminates the risk of fracture, with 4 development points ensuring precise implantation, and nickel titanium alloy material providing excellent biocompatibility. 02 Excellent durability: validated through high standards and rigorous testing The extreme fatigue test simulating 10 years of human movement, including hundreds of millions of compressions, tens of millions of bending and twisting tests, showed that the bracket did not break, proving its long-term reliability.
03 Complete specifications: meet diverse clinical needs Provide a variety of diameters, lengths, and carving segment specifications, fully covering clinical anatomical needs, supporting personalized treatment plans, and reducing intraoperative splicing to improve surgical efficiency。 04 Excellent clinical data: confirmed excellent therapeutic effect The results of multicenter clinical trials showed that, V-Mixtent ® The performance of the venous stent is excellent. Clinical studies have shown that the patency rate of thrombotic type is over 82% one year after surgery, while non thrombotic type can reach 97.92%. After 3 years of surgery, the patency rate of thrombotic type remains close to 78%, while non thrombotic type can reach 93.61%. Ensheng Medical V-Mixtent ® The research and application of venous stents is an important achievement of Ensheng Medical's continuous deepening of the field of venous treatment, focusing on the combination of technological innovation and clinical needs. With V-Mixtent ® Further widespread clinical application is expected to provide high-quality treatment options for more patients with venous diseases, while also contributing to the sustained development of China's medical equipment industry. The exhibition booth of Ensheng Medical attracted the interest of many attending Chinese and foreign doctors, who had a detailed understanding of V-Mixtent ® The technical characteristics and clinical advantages of intravenous stents. Many doctors have shown great concern for the modular design of products and have conducted in-depth discussions with on-site technicians on product specification selection and clinical operation details. Shanghai Ensheng Medical Technology Co., Ltd. was established in November 2018, integrating research and development with production. It is located in the Lingang New Area of China (Shanghai) Pilot Free Trade Zone. Ensheng Medical is a high-tech medical innovation enterprise with over 100 independent intellectual property rights. It focuses on the research and industrialization of advanced vascular medical devices at the international level. Based on three platforms: modular design of stents, biomedical materials, and active medical equipment, it creates a comprehensive solution for peripheral venous disease and is committed to bringing more high-end medical devices that benefit the public to clinical doctors and patients. The company attracts numerous high-quality talents to join, with talents in various fields from research and development production to clinical registration. It is equipped with a 2300 square meter enterprise office building and a 1000 square meter GMP 10000 level purification workshop, and has passed the ISO13485 international standard system certification.
|